BIOMOLECULES

CARBOHYDRATE
*The structure of sugars was first elucidated by Emil Fischer, a German chemist.
2. Disaccharides, the double sugar formed by two monosaccharides joined by a covalent
bonds.
3. Polysaccharides, polymers composed of many sugar building blocks.
*The structure of sugars was first elucidated by Emil Fischer, a German chemist.
- Carbohydrate include both sugar and polymers of sugar.
- The three forms of carbohydrate categorized based on its complexity :
2. Disaccharides, the double sugar formed by two monosaccharides joined by a covalent
bonds.
3. Polysaccharides, polymers composed of many sugar building blocks.
MONOSACCHARIDES.
- The simples form of sugar.
- The general molecular formula is CnH2nOn.
- Glucose is the most common type of monosaccharides.
- The general structure of glucose in linear and ring form are :
- 2 different types of functional groups in glucose :
- Carbonyl (C=O)
- Multiple hydroxyl groups (-OH)
DISACCHARIDES
- Disaccharides consist of two monosaccharides which is joined by glycosidic linkage.
- Glycosidic linkage is a covalent bond formed by dehydration reaction.
- Two glucose molecules linked together to form a maltose.
- Glucose and fructose linked together to form sucrose by dehydration process
- The general structure of disaccharides are :
POLYSACCHARIDES
- Polysaccharides are polymer with many monosaccharides joined by glycosidic linkage.
- Different forms of polysaccharides include :
- Storage polysaccharides (starch and glycogen)
- Structural polysaccharides (cellulose)
- The general structure of polysaccharides are :
- Starch is a polymer formed by alpha glucose joined by 1-4 glycosidic linkage.
Carbohydrate metabolism.
Carbohydrate metabolism involve 4 process :
1. glycolysis
2. gluconeogenesis
3. glycogenesis
4. glycogenolysis
GLYCOLYSIS
Glycolysis, part of cellular respiration, is a series of reactions that constitute the first phase of most carbohydrate catabolism. Catabolism meaning the breaking down of larger molecules into smaller ones. The word glycolysis is derived from two Greek words and means the breakdown of something sweet. Glycolysis breaks down glucose and forms pyruvate with the production of two molecules of ATP. The pyruvate end product of glycolysis can be used in either anaerobic respiration if no oxygen is available or in aerobic respiration via the TCA cycle which yields much more usable energy for the cell. The process of glycolysis is occurs in cytosol of the cells.
Carbohydrate metabolism involve 4 process :
1. glycolysis
2. gluconeogenesis
3. glycogenesis
4. glycogenolysis
GLYCOLYSIS
Glycolysis, part of cellular respiration, is a series of reactions that constitute the first phase of most carbohydrate catabolism. Catabolism meaning the breaking down of larger molecules into smaller ones. The word glycolysis is derived from two Greek words and means the breakdown of something sweet. Glycolysis breaks down glucose and forms pyruvate with the production of two molecules of ATP. The pyruvate end product of glycolysis can be used in either anaerobic respiration if no oxygen is available or in aerobic respiration via the TCA cycle which yields much more usable energy for the cell. The process of glycolysis is occurs in cytosol of the cells.
GLUCONEOGENESIS
Gluconeogenesis is a metabolic pathway that results in the generation of glucose from non-carbohydrate carbon substrates such as pyruvate, lactate, glycerol, and glucogenic amino acids.
It is one of the two main mechanisms used by humans and many other animals to maintain blood glucose levels and avoiding low blood glucose level (hypoglycemia).
Gluconeogenesis is a ubiquitous process, present in plants, animals, fungi, bacteria, and other microorganisms. In vertebrates, gluconeogenesis takes place mainly in the liver and, to a lesser extent, in the cortex of the kidneys. In ruminants, this tends to be a continuous process. In many other animals, the process occurs during periods of fasting, starvation, low-carbohydrate diets, or intense exercise. The process is highly endergonic until it is coupled to the hydrolysis of ATP or GTP, effectively making the process exergonic. For example, the pathway leading from pyruvate to glucose-6-phosphate requires 4 molecules of ATP and 2 molecules of GTP to proceed spontaneously. Gluconeogenesis is often associated with ketosis. Gluconeogenesis is also a target of therapy for type 2 diabetes, such as the antidiabetic drug, metformin, which inhibits glucose formation and stimulates glucose uptake by cells. In ruminants, because metabolizable dietary carbohydrates tend to be metabolized by rumen organisms. Gluconeogenesis occurs regardless of fasting, low-carbohydrate diets and exercise.
Gluconeogenesis is a metabolic pathway that results in the generation of glucose from non-carbohydrate carbon substrates such as pyruvate, lactate, glycerol, and glucogenic amino acids.
It is one of the two main mechanisms used by humans and many other animals to maintain blood glucose levels and avoiding low blood glucose level (hypoglycemia).
Gluconeogenesis is a ubiquitous process, present in plants, animals, fungi, bacteria, and other microorganisms. In vertebrates, gluconeogenesis takes place mainly in the liver and, to a lesser extent, in the cortex of the kidneys. In ruminants, this tends to be a continuous process. In many other animals, the process occurs during periods of fasting, starvation, low-carbohydrate diets, or intense exercise. The process is highly endergonic until it is coupled to the hydrolysis of ATP or GTP, effectively making the process exergonic. For example, the pathway leading from pyruvate to glucose-6-phosphate requires 4 molecules of ATP and 2 molecules of GTP to proceed spontaneously. Gluconeogenesis is often associated with ketosis. Gluconeogenesis is also a target of therapy for type 2 diabetes, such as the antidiabetic drug, metformin, which inhibits glucose formation and stimulates glucose uptake by cells. In ruminants, because metabolizable dietary carbohydrates tend to be metabolized by rumen organisms. Gluconeogenesis occurs regardless of fasting, low-carbohydrate diets and exercise.
GLYCOLYSIS VS GLUCONEOGENESIS
|
GLYCOLYSIS
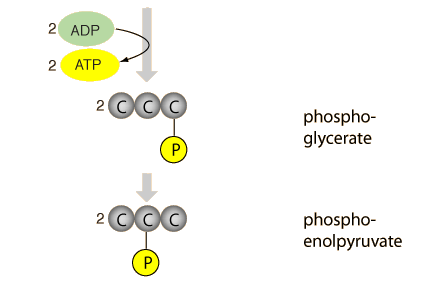
In glycolysis the following steps are irreversible: a) Glucose to Glucose 6-phosphate b) Fructose 6-phosphate to Fructose 1,6-bisphosphate c) Phosphoenolpyruvate to Pyruvate |
GLUCONEOGENESIS
In gluconeogenesis those three irreversible steps are bypassed by the following reactions: a) Glucose 6-phosphate + H2O ----> glucose + Pi (catalysed by the enzyme glucose 6-phosphatase) b) Fructose 1,6-bisphosphate + H2O ----> fructose 6-phosphate + Pi (catalysed by the enzyme fructose 1,6-bisphosphatase) c) Pyruvate + CO2 + ATP + H2O ----> oxaloacetate + ADP + Pi + 2H+ (catalysed by the enzyme pyruvate carboxylase) Oxaloacetate + GTP <==> phosphoenolpyruvate + GDP + CO2 (catalysed by the enzyme phosphoenol carboxykinase). The gluconeogenesis take place in the cytosol and partly in the mitochondrial matrix. |
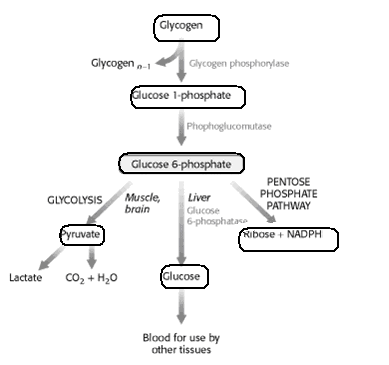
GLYCOGENESIS
Glycogenesis is the formation of glycogen from glucose. Glycogen is synthesized depending on the demand for glucose and ATP (energy). If both are present in relatively high amounts, then the excess of insulin promotes the glucose conversion into glycogen for storage in liver and muscle cells.
In the synthesis of glycogen, one ATP is required per glucose incorporated into the polymeric branched structure of glycogen. actually, glucose-6-phosphate is the cross-roads compound. Glucose-6-phosphate is synthesized directly from glucose or as the end product of gluconeogenesis.
GLYCOGENOLYSIS
Glycogenolysis is the process of glycogen breakdown into glucose. In glycogenolysis, glycogen stored in the liver and muscles, is converted first to glucose-1- phosphate and then into glucose-6-phosphate. Two hormones which control glycogenolysis are a peptide, glucagon from the pancreas and epinephrine from the adrenal glands.
Glucagon is released from the pancreas in response to low blood glucose and epinephrine is released in response to a threat or stress. Both hormones act upon enzymes to stimulate glycogen phosphorylase to begin glycogenolysis and inhibit glycogen synthetase (to stop glycogenesis).
Glycogen is a highly branched polymeric structure containing glucose as the basic monomer. First individual glucose molecules are hydrolyzed from the chain, followed by the addition of a phosphate group at C-1. In the next step the phosphate is moved to the C-6 position to give glucose 6-phosphate, a cross road compound.
Glucose-6-phosphate is the first step of the glycolysis pathway if glycogen is the carbohydrate source and further energy is needed. If energy is not immediately needed, the glucose-6-phosphate is converted to glucose for distribution in the blood to various cells such as brain cells.
GLYCOGENESIS VS GLYCOGENOLYSIS
PENTOSE PHOSPHATE PATHWAY
In biochemistry, the pentose phosphate pathway is a metabolic pathway parallel to glycolysis that generates NADPH and pentoses (5-carbon sugars). While it does involve oxidation of glucose, its primary role is anabolic rather than catabolic.
There are two distinct phases in the pathway. The first is the oxidative phase, in which NADPH is generated, and the second is the non-oxidative synthesis of 6-carbon sugars. For most organisms, the pentose phosphate pathway takes place in the cytosol; in plants, most steps take place in plastids.
PROTEIN
- Proteins account for more than 50% of dry mass of most cells.
- Amino acid is the simplest form of protein.
- Amino acid is an organic molecule possessing both amino group (- NH2), carboxyl group (- COOH), a hydrogen atom and a variable group symbolized by R.
- The R group or side chain differ with each amino acid. The side chain could be as simple as a hydrogen atom (as in glycine) or with various functional group (as in glutamine).
- Amino acids are grouped according to the properties of side chain (R-group):
- Non-polar side chain which is hydrophobic.
- Polar side chain which is hydrophilic.
- Carboxyl side chain which is generally negative in charge categorize as acidic amino acids.
- Amino side chain which is generally positive in charge categorize as basic amino acids.
Classes of amino acid
There are 20 different amino acids most commonly occurring in nature. Each of them has its specific characteristics defined by the side chain, which provides it with its unique role in a protein structure. Based on the propensity of the side chain to be in contact with polar solvent like water, it may be classified ashydrophobic (low propensity to be in contact with water), polar or charged (energetically favorable contact with water).
There are basically four different classes of amino acids determined by different side chains: (1) non-polar and neutral, (2) polar and neutral, (3) acidic and polar, (4) basic and polar.
There are 20 different amino acids most commonly occurring in nature. Each of them has its specific characteristics defined by the side chain, which provides it with its unique role in a protein structure. Based on the propensity of the side chain to be in contact with polar solvent like water, it may be classified ashydrophobic (low propensity to be in contact with water), polar or charged (energetically favorable contact with water).
There are basically four different classes of amino acids determined by different side chains: (1) non-polar and neutral, (2) polar and neutral, (3) acidic and polar, (4) basic and polar.
Polypeptide chain
- Polypeptides are polymers of amino acids.
- Polypeptides are formed when two amino acids are positioned so that the carboxyl group (- COOH) of one is adjacent to the amino group (-HN2) of the other, followed by a dehydration reaction (removal of water molecule) and joined by a covalent bond called peptide bond.
Function of amino acid
Amino acids build proteins, and proteins are life-sustaining macronutrients. Yet simply calling amino acids the building blocks of protein doesn’t do justice to their value. While some amino acids only make proteins, others fill a variety of roles, from supporting metabolism to protecting human heart. Our body can also use amino acids for energy when we lack carbohydrates and fats.
Nitrogen Metabolism.
Nitrogen metabolism is about the flow of nitrogen cycle. The nitrogen cycle describes how nitrogen moves between plants, animals, bacteria, the air in atmosphere , and soil in the ground. Nitrogen is an important element to all life on Earth.
Different Nitrogen States
For Nitrogen to be used by different life forms on Earth, it must change into different states. Nitrogen in the atmosphere, or air, is N2. Other important states of nitrogen include Nitrates (N03), Nitrites (NO2), and Ammonium (NH4).
Nitrogen Cycle
This picture shows the flow of the nitrogen cycle. The most important part of the cycle is bacteria. Bacteria help the nitrogen change between states so it can be used. When nitrogen is absorbed by the soil, different bacteria help it to change states so it can be absorbed by plants. Animals then get their nitrogen from the plants.
Amino acids build proteins, and proteins are life-sustaining macronutrients. Yet simply calling amino acids the building blocks of protein doesn’t do justice to their value. While some amino acids only make proteins, others fill a variety of roles, from supporting metabolism to protecting human heart. Our body can also use amino acids for energy when we lack carbohydrates and fats.
Nitrogen Metabolism.
Nitrogen metabolism is about the flow of nitrogen cycle. The nitrogen cycle describes how nitrogen moves between plants, animals, bacteria, the air in atmosphere , and soil in the ground. Nitrogen is an important element to all life on Earth.
Different Nitrogen States
For Nitrogen to be used by different life forms on Earth, it must change into different states. Nitrogen in the atmosphere, or air, is N2. Other important states of nitrogen include Nitrates (N03), Nitrites (NO2), and Ammonium (NH4).
Nitrogen Cycle
This picture shows the flow of the nitrogen cycle. The most important part of the cycle is bacteria. Bacteria help the nitrogen change between states so it can be used. When nitrogen is absorbed by the soil, different bacteria help it to change states so it can be absorbed by plants. Animals then get their nitrogen from the plants.
Amino acid Anabolism
Anabolism is the process by which the body utilizes the energy released by catabolism to synthesize complex molecules. These complex molecules are then utilized to form cellular structures that are formed from small and simple precursors that act as building blocks.
Amino acids are synthesized from the carbon skeleton (derived from citric acid cycle intermediates) and from amino groups from glutamate or glutamine. We can synthesize 11 amino acids (nonessential). The other 9 are considered essential: H, I, L, K, M, F, T, W, V. From these 9, some we cannot produce, others we can produce just not enough. Arginine is essential in children but adults can produce enough.
Transamination
Most amino acids are deaminated by transamination (or transfer of amino group), a chemical reaction that transfer their amino group to an keto-acid forming new amino acids.
Most standard amino acids lose their α-amino group early on in degradation through transamination, that is, transfer to an α-keto acid. The diagram above is illustrated for alanine, which transfers its amino group to α-ketoglutarate to become pyruvate. Transamination is mediated by several different aminotransferase enzymes. These may be specific for individual amino acids, or they may be able to process a group of chemically similar ones. The latter applies to the group of the branched-chain amino acids, which comprises leucine, isoleucine, and valine.
In the case of alanine, the α-keto acid that accepts the amino group is α-ketoglutarate; this also applies to most other amino acids. Transamination is freely reversible; therefore, both glutamate and α-ketoglutarate are substrates of multiple transaminases. If amino groups are to be transferred between two amino acids other than glutamate, this will usually involve the formation of glutamate as an intermediate.
In the case of alanine, the α-keto acid that accepts the amino group is α-ketoglutarate; this also applies to most other amino acids. Transamination is freely reversible; therefore, both glutamate and α-ketoglutarate are substrates of multiple transaminases. If amino groups are to be transferred between two amino acids other than glutamate, this will usually involve the formation of glutamate as an intermediate.
Amino acid Catabolism
Protein catabolism is the breakdown of proteins into amino acid and simple derivative compounds, for transport into the cell through the plasma membrane and ultimately for the polymerisation into new proteins via the use of ribonucleic acids (RNA) and ribosomes. Protein catabolism, which is the breakdown of macromolecules, is essentially a digestion process.
The amino acids produced by catabolism may be directly recycled, used to make new amino acids, or undergo amino acid catabolism to be converted to other compounds via the Kreb cycles.
Protein catabolism is the breakdown of proteins into amino acid and simple derivative compounds, for transport into the cell through the plasma membrane and ultimately for the polymerisation into new proteins via the use of ribonucleic acids (RNA) and ribosomes. Protein catabolism, which is the breakdown of macromolecules, is essentially a digestion process.
The amino acids produced by catabolism may be directly recycled, used to make new amino acids, or undergo amino acid catabolism to be converted to other compounds via the Kreb cycles.
Amino acids must first pass out of organelles and cells into blood circulation via amino acid transporters, since the amine and carboxylic acid groups are typically ionized. Degradation of an amino acid, occurring in the liver and kidneys, often involves deamination by moving its amino group to alpha-ketoglutarate, forming glutamate. This process involves transaminases, often the same as those used in amination during synthesis. In many vertebrates, the amino group is then removed through the urea cycle and is excreted in the form of urea.
Catabolism of proteinogenic amino acids. Amino acids can be classified according to the properties of their main products as either of the following:
* Glucogenic, with the products having the ability to form glucose by gluconeogenesis
* Ketogenic, with the products not having the ability to form glucose. These products may still be used for ketogenesis or lipid synthesis.
* Amino acids catabolized into both glucogenic and ketogenic products.
* Glucogenic, with the products having the ability to form glucose by gluconeogenesis
* Ketogenic, with the products not having the ability to form glucose. These products may still be used for ketogenesis or lipid synthesis.
* Amino acids catabolized into both glucogenic and ketogenic products.
LIPIDS
b) phopspholipid
c) steroids
- Lipids are not true polymers.
- They are generally not big enough to be considered as macromolecules.
- They mix poorly with water.
- There are three important types of lipid :
b) phopspholipid
c) steroids
Fats/ Triacylglycerol
- Fats are not polymers, they are large molecules assembled from smaller molecules by dehydration reactions.
- A fat or triglyceride is constructed by one molecule of glycerol and three molecules of fatty acids.
- Three fatty acids and a glycerol is joined by an ester linkage, a bond between hydroxyl group (-OH) and carboxyl group (- COOH).
- Fatty acid comprises a long carbon skeleton (16 to 18 carbon atoms), at one end of the carbon skeleton is a part of carboxyl group (-COOH). The rest of skeleton consists of hydrocarbon chain. This hydrocarbon (C – H) of fatty acid is relatively non-polar, and is said to be hydrophobic.
- Glycerol is an alcohol. Each of its three carbon bears a hydroxyl group (- OH).
Phospholipids
- Phospholipids are an important component of cell as they make up of the cell membrane.
- The structure of a phospholipid comprises of one molecule of glycerol joined with two molecules of fatty acids. The third hydroxyl group of glycerol is joined with a phosphate group, which has a negative electric charge in the cell.
Steroids
- The structure of steroid comprises four carbon skeleton rings fused together.
LIPID METABOLISM
FATTY ACID DEGRADATION
Fatty acid degradation is the process in which fatty acids are broken down into their metabolites, in the end generating acetyl-CoA, the entry molecule for the citric acid cycle, the main energy supply of animals. It includes three major steps:
1. Lipolysis of and release from adipose tissue
2. Activation and transport into mitochondria
3. β-oxidation
Lipolysis and release from adipose tissues
Initially in the process of degradation, fatty acids are stored in fat cells (adipocytes). The breakdown of this fat is known as lipolysis. The products of lipolysis, free fatty acids, are released into the bloodstream and circulate throughout the body. During the breakdown of triacylglycerols into fatty acids, more than 75% of the fatty acids are reconverted to triacylglycerol, even in cases of starvation and exercise.
Activation and transport into mitochondria
Fatty acids must be activated before they can be carried into the mitochondria, where fatty acid oxidation occurs. This process occurs in two steps catalyzed by the enzyme fatty acyl-CoA synthetase.
The inner mitochondrial membrane is impermeable to fatty acids and a specialized carnitine carrier system operates to transport activated fatty acids from cytosol to mitochondria.
Once activated, the acyl CoA is transported into the mitochondrial matrix. This occurs via a series of similar steps:
1. Acyl CoA is conjugated to carnitine by carnitine acyltransferase I (palmitoyltransferase I )
located on the outer mitochondrial membrane
2. Acyl carnitine is shuttled inside by a translocase
3.Acyl carnitine (such as Palmitoylcarnitine) is converted to acyl CoA by carnitine
acyltransferase (palmitoyltransferase) II located on the inner mitochondrial membrane. The
liberated carnitine returns to the cytosol.
Once inside the mitochondria, the β-oxidation of fatty acids occurs via five recurring steps:
1. Activation by ATP
2. Oxidation by FAD,
3. Hydration,
4. Oxidation by NAD+,
5. Thiolysis,
6. The final product is acetyl-CoA, the entry molecule for the citric acid cycle.
FATTY ACID DEGRADATION
Fatty acid degradation is the process in which fatty acids are broken down into their metabolites, in the end generating acetyl-CoA, the entry molecule for the citric acid cycle, the main energy supply of animals. It includes three major steps:
1. Lipolysis of and release from adipose tissue
2. Activation and transport into mitochondria
3. β-oxidation
Lipolysis and release from adipose tissues
Initially in the process of degradation, fatty acids are stored in fat cells (adipocytes). The breakdown of this fat is known as lipolysis. The products of lipolysis, free fatty acids, are released into the bloodstream and circulate throughout the body. During the breakdown of triacylglycerols into fatty acids, more than 75% of the fatty acids are reconverted to triacylglycerol, even in cases of starvation and exercise.
Activation and transport into mitochondria
Fatty acids must be activated before they can be carried into the mitochondria, where fatty acid oxidation occurs. This process occurs in two steps catalyzed by the enzyme fatty acyl-CoA synthetase.
The inner mitochondrial membrane is impermeable to fatty acids and a specialized carnitine carrier system operates to transport activated fatty acids from cytosol to mitochondria.
Once activated, the acyl CoA is transported into the mitochondrial matrix. This occurs via a series of similar steps:
1. Acyl CoA is conjugated to carnitine by carnitine acyltransferase I (palmitoyltransferase I )
located on the outer mitochondrial membrane
2. Acyl carnitine is shuttled inside by a translocase
3.Acyl carnitine (such as Palmitoylcarnitine) is converted to acyl CoA by carnitine
acyltransferase (palmitoyltransferase) II located on the inner mitochondrial membrane. The
liberated carnitine returns to the cytosol.
Once inside the mitochondria, the β-oxidation of fatty acids occurs via five recurring steps:
1. Activation by ATP
2. Oxidation by FAD,
3. Hydration,
4. Oxidation by NAD+,
5. Thiolysis,
6. The final product is acetyl-CoA, the entry molecule for the citric acid cycle.
FATTY ACID BIOSYNTHESIS
• Synthesis takes place in the cytosol
• Intermediates covalently linked to acyl carrier protein
• Activation of each acetyl CoA.
• acetyl CoA + CO2 = Malonyl CoA
• Four-step repeating cycle, extension by 2-carbons / cycle
– Condensation
– Reduction
– Dehydration
– reduction
• Synthesis takes place in the cytosol
• Intermediates covalently linked to acyl carrier protein
• Activation of each acetyl CoA.
• acetyl CoA + CO2 = Malonyl CoA
• Four-step repeating cycle, extension by 2-carbons / cycle
– Condensation
– Reduction
– Dehydration
– reduction
Palmitate is the end product released by fatty acid synthase; fatty acids with longer chains are formed from palmitate by separate elongase enzymes. Inside the mitochondria, the pathway of elongation is mostly a reversal of β-oxidation, except that NADPH is used instead of FADH2 in the final reduction of enoyl-CoA. In the ER, the chemistry resembles that of fatty acid synthase, except that the substrate is bound to coenzyme A rather than to the enzyme itself.
CHOLESTROL SYNTHESIS
The process of cholestrol synthesis can be considered to be composed of five major steps :-
1. Acetyl-CoA are converted to 3-hydroxy-3-methylglutaryl-CoA (HMG-CoA)
2. HMG-CoA is converted to mevalonate
3. Mevalonate is converted to the isoprene based molecule, isopentenyl pyrophosphate (IPP) with the concomitant loss of carbon dioxide
4. IPP is converted to squalene
5. Squalene is converted to cholestrol
The process of cholestrol synthesis can be considered to be composed of five major steps :-
1. Acetyl-CoA are converted to 3-hydroxy-3-methylglutaryl-CoA (HMG-CoA)
2. HMG-CoA is converted to mevalonate
3. Mevalonate is converted to the isoprene based molecule, isopentenyl pyrophosphate (IPP) with the concomitant loss of carbon dioxide
4. IPP is converted to squalene
5. Squalene is converted to cholestrol
Transportation of lipid in bloods.
Blood lipids (or blood fats) are lipids in the blood, either free or bound to other molecules. They are mostly transported in a protein capsule, and the density of the lipids and type of protein determines the fate of the particle and its influence on metabolism. The concentration of blood lipids depends on intake and excretion from the intestine, and uptake and secretion from cells. Blood lipids are mainly fatty acids[citation needed] and cholesterol. Hyperlipidemia is the presence of elevated or abnormal levels of lipids and/or lipoproteins in the blood, and is a major risk factor for cardiovascular disease.
Fatty acid and cholesterol transport in plasma lipoproteins evolved in the context of an open circulatory system in which lipoprotein particles are secreted directly into the blood and have ready access to cells in various tissues. In higher vertebrates with closed capillary beds, hydrolysis of triglycerides at capillary surfaces is required for efficient uptake of their component fatty acids into cells. Likewise, hydrolysis of cellular triglycerides in cells of adipose tissue precedes mobilization of the fatty acids and permits large amounts to be transported in the blood. However, in all Metazoa lipoproteins are secreted primarily from cells adjacent to an open microvascular bed. Uptake of lipoprotein particles as such into cells occurs in invertebrates and vertebrates alike, facilitated by binding to high-affinity receptors on cell surfaces. In vertebrates, a concentration gradient created between cholesterol in cells and lipoproteins by a cholesterol-esterifying enzyme that acts on lipoproteins promotes movement of cholesterol into the plasma compartment. Thus the strategies to transport poorly soluble lipids include enzymatic reactions at cell surfaces and in blood plasma as well as the processes of exocytosis and endocytosis.
NUCLEIC ACIDS
- Gene is the basic unit of inheritance.
- Gene consists of DNA, which belongs to a class compound called nucleic acids.
- Nucleic acids are polymers made of monomers called nucleotides.
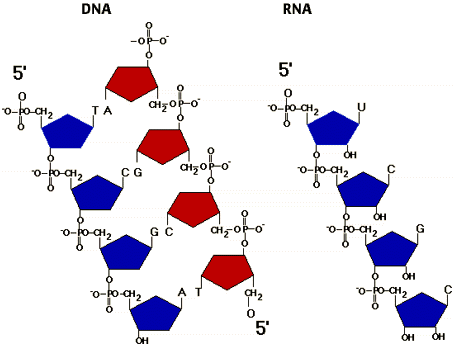
- There are two types of nucleic acids, DNA (deoxyribonucleic acid) and RNA (ribonucleic acid).
- Nucleic acids are macromolecules that exists as polymers called polynucleotides.
- Polynucleotide consists of monomers called nucleotides.
- A nucleotide is composed of three parts:
- Five- carbon sugar (pentose sugar).
- A nitrogenous base.
- A phosphate group.
Nucleotide Polymers
- Nucleotides are joined by phosphodiester linkage, which consists of a phosphate group links to sugars of two nucleotides.
- One end has a phosphate attached to a 5’ carbon, the other end has a hydroxyl group on a 3’ carbon.
- This bonding results in a backbone with a repeating pattern of sugar-phosphate units (sugar-phosphate backbones).
AEROBIC METABOLISM
CONVERSION OF PYRUVATE TO ACETYL-COA
In order for pyruvate, the product of glycolysis, to enter the next pathway, it must undergo several changes to become acetyl Coenzyme A (acetyl CoA). Acetyl CoA is a molecule that is further converted to oxaloacetate, which enters the citric acid cycle (Krebs cycle). The conversion of pyruvate to acetyl CoA is a three-step process .
CONVERSION OF PYRUVATE TO ACETYL-COA
In order for pyruvate, the product of glycolysis, to enter the next pathway, it must undergo several changes to become acetyl Coenzyme A (acetyl CoA). Acetyl CoA is a molecule that is further converted to oxaloacetate, which enters the citric acid cycle (Krebs cycle). The conversion of pyruvate to acetyl CoA is a three-step process .
Step 1. A carboxyl group is removed from pyruvate, releasing a molecule of carbon dioxide into the surrounding medium. (Note: carbon dioxide is one carbon attached to two oxygen atoms and is one of the major end products of cellular respiration. ) The result of this step is a two-carbon hydroxyethyl group bound to the enzyme pyruvate dehydrogenase; the lost carbon dioxide is the first of the six carbons from the original glucose molecule to be removed. This step proceeds twice for every molecule of glucose metabolized (remember: there are two pyruvate molecules produced at the end of glycolysis); thus, two of the six carbons will have been removed at the end of both of these steps.
Step 2. The hydroxyethyl group is oxidized to an acetyl group, and the electrons are picked up by NAD+, forming NADH (the reduced form of NAD+). The high-energy electrons from NADH will be used later by the cell to generate ATP for energy.
Step 3. The enzyme-bound acetyl group is transferred to CoA, producing a molecule of acetyl CoA. This molecule of acetyl CoA is then further converted to be used in the next pathway of metabolism, the citric acid cycle.
REACTION OF CITRIC ACID CYCLE
As the above diagram illustrates, this is truly a continuous cycle. Acetyl coenzyme A binds to the oxaloacetate in order to convert it to citrate. Water is released from the citrate and then regained in order to form isocitrate. NAD+ binds to isocitrate, leaves as NADH, and carbon dioxide is lost in order to form a-ketoglutarate. NAD+ binds to a-ketoglutarate, leaves as NADH, coenzyme A binds and carbon dioxide is lost in order to form succinyl CoA. Coenzyme A leaves in order to form succinate. FAD binds to succinate and leaves as FADH2 in order to form fumarate. Water binds to fumarate and forms malate. NAD binds to malate and leaves as NADH which creates a new oxaloacetate molecule ready to bind to another acetyl coenzyme A to start the process over again.
Step 2. The hydroxyethyl group is oxidized to an acetyl group, and the electrons are picked up by NAD+, forming NADH (the reduced form of NAD+). The high-energy electrons from NADH will be used later by the cell to generate ATP for energy.
Step 3. The enzyme-bound acetyl group is transferred to CoA, producing a molecule of acetyl CoA. This molecule of acetyl CoA is then further converted to be used in the next pathway of metabolism, the citric acid cycle.
REACTION OF CITRIC ACID CYCLE
As the above diagram illustrates, this is truly a continuous cycle. Acetyl coenzyme A binds to the oxaloacetate in order to convert it to citrate. Water is released from the citrate and then regained in order to form isocitrate. NAD+ binds to isocitrate, leaves as NADH, and carbon dioxide is lost in order to form a-ketoglutarate. NAD+ binds to a-ketoglutarate, leaves as NADH, coenzyme A binds and carbon dioxide is lost in order to form succinyl CoA. Coenzyme A leaves in order to form succinate. FAD binds to succinate and leaves as FADH2 in order to form fumarate. Water binds to fumarate and forms malate. NAD binds to malate and leaves as NADH which creates a new oxaloacetate molecule ready to bind to another acetyl coenzyme A to start the process over again.
ELECTRON TRANSPORT CHAIN & FUNCTION
The electron transport chain is the final component of aerobic respiration and is the only part of glucose metabolism that uses atmospheric oxygen . Electron transport is a series of redox reactions that resemble a relay race in that electrons are passed rapidly from one component to the next, to the endpoint of the chain where the electrons reduce molecular oxygen, producing water. There are four complexes composed of proteins, labeled I through IV; the aggregation of these four complexes, together with associated mobile accessory electron carriers, is called the electron transport chain. The electron transport chain is present in multiple copies in the inner mitochondrial membrane of eukaryotes and the plasma membrane of prokaryotes.
During various steps in glycolysis and the citric acid cycle, the oxidation of certain intermediate precursor molecules causes the reduction of NAD+ to NADH + H+ and FAD to FADH2. NADH and FADH2 then transfer protons and electrons to the electron transport chain to produce additional ATPs from oxidative phosphorylation.
REDUCTION POTENTIAL
Reduction potential (also known as redox potential, oxidation / reduction potential, ORP, pE, ε, or ) is a measure of the tendency of a chemical species to acquire electrons and thereby be reduced.
OXIDATIVE PHOSPHORYLATION.
Oxidative phosphorylation is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP.
CHEMIOSMOTIC THEORY AND ATP SYNTHESIS
The chemiosmotic theory explains how ATP is generated in the mitochondria via the electron transfer chain (ETC). ETC is an oxidative phosphorylation reaction takes place in the inner membrane of the mitochondria. It begins with NADH and FADH2 that looses electrons. These electrons are transferred along the chain. As each complex accepts electrons and passes it on, energy is released. This energy is used to pump the protons move against the concentration gradient from the matrix of the mitochondria to the inner membrane space. Eventually, there is a high concentration of protons build up the membrane space and the protons try to move back into the matrix. However, the inner membrane is impermeable to protons. A proton motive force (PMF) is set up and ATP synthase undergoes conformational change and uses the PMF to make ATP from ADP. ATP synthase allows the proton to diffuse back down their gradient.
ATP + Pi ——-> ATP
The chemiosmotic theory explains how ATP is generated in the mitochondria via the electron transfer chain (ETC). ETC is an oxidative phosphorylation reaction takes place in the inner membrane of the mitochondria. It begins with NADH and FADH2 that looses electrons. These electrons are transferred along the chain. As each complex accepts electrons and passes it on, energy is released. This energy is used to pump the protons move against the concentration gradient from the matrix of the mitochondria to the inner membrane space. Eventually, there is a high concentration of protons build up the membrane space and the protons try to move back into the matrix. However, the inner membrane is impermeable to protons. A proton motive force (PMF) is set up and ATP synthase undergoes conformational change and uses the PMF to make ATP from ADP. ATP synthase allows the proton to diffuse back down their gradient.
ATP + Pi ——-> ATP
ATP SYNTHASE
ATP synthase is the enzyme that makes ATP by chemiosmosis. It allows protons to pass through the membrane and uses the kinetic energy to phosphorylate ADP, making ATP. The generation of ATP by chemiosmosis occurs in chloroplasts and mitochondria as well as in most bacteria and archaea.
THE CHEMIOSMOTIC THEORY OF GENERATION OF ATP AS A RESULT OF AN ELECTRON TRANSPORT CHAIN.
According to this theory, the tranfer of electrons down an electron transport system through a series of oxidation-reduction reactions releases energy. This energy allows certain carriers in the chain to transport hydrogen ions (H+ or protons) across a membrane. The fluid on the side of the membrane where the protons accumulate acquires a positive charge; the fluid on the opposite side of the membrane is left with a negative charge. The energized state of the membrane as a result of this charge separation is called proton motive force or PMF and provides the energy necessary for enzymes called ATP synthases to catalyze the synthesis of ATP from ADP and phosphate. This generation of ATP occurs as the protons cross the membrane through the ATP synthase complexes and re-enter either the bacterial cytoplasm or the matrix of the mitochondria.
COMPARISON WHERE THE ELECTRON TRANSPORT CHAIN OCCURS IN PROKARYOTIC CELLS AND IN EUKARYOTIC CELLS.
Depending on the type of cell, the electron transport chain may be found in the cytoplasmic membrane or the inner membrane of mitochondria.
In prokaryotic cells, the protons are transported from the cytoplasm of the bacterium across the cytoplasmic membrane to the periplasmic space located between the cytoplasmic membrane and the cell wall .
In eukaryotic cells, protons are transported from the matrix of the mitochondria across the inner mitochondrial membrane to the intermembrane space located between the inner and outer mitochondrial membranes.
COMPARE AND CONTRAST BETWEEN MALATE-ASPARTATE SHUTTLE AND GLYCEROL-3-PHOSPHATE SHUTTLE
|
MALATE-ASPARTATE SHUTTLE
1. The malate-aspartate shuttle is a biochemical system for translocating electrons produced during glycolysis across the semipermeable inner membrane of the mitochondrion for oxidative phosphorylation in eukaryotes. These electrons enter the electron transport chain of the mitochondria via reduction equivalents to generate ATP. 2. The shuttle system is required because the mitochondrial inner membrane is impermeable to NADH, the primary reducing equivalent of the electron transport chain. To circumvent this, malate carries the reducing equivalents across the membrane. 3.Malate-Aspartate Shuttle make 3 ATP. 4.Location of process is at heart and liver. |
GLYCEROL-3-PHOSPHATE SHUTTLE
1. The glycerol-3-phosphate shuttle is a mechanism that regenerates NAD+ from NADH, a by-product of glycolysis. Its importance in transporting reducing equivalents is secondary to the malate-aspartate shuttle. 2. The glycerol-3-phosphate shuttle allows the NADH synthesized in the cytosol by glycolysis to contribute to the oxidative phosphorylation pathway in the mitochondria to generate ATP.It has been found in animals, fungi, and plants. 3. G3P shuttle make 2 ATP . 4. The location of process is at skeletal muscle and brain. |
METABOLIC PROFILES IN BRAIN, SKELETAL MUSCLES, HEART MUSCLES, LIVER AND ADIPOSE TISSUES.
BRAIN
SKELETAL MUSCLES.
METABOLIC CONTROL BY HORMONE
The metabolic pathways are complex and interdependent. With the changing environments the reactions of metabolism must be finely regulated to maintain a constant set of conditions within cells, a condition called homeostasis. Control of metabolic pathways also allows organisms to respond to signals and interact actively with their environments.
Control of metabolism
Coarse Control: control of the amount of an enzyme. This is a slow process as it involves protein
synthesis.
Fine Control: control of the activity of the enzyme. This is a fast process as it involves changing
the activity of enzyme already available in the cells.
For example :, the metabolic control of carbohydrate.
Glucose homeostasis is a complicated interaction of metabolic pathways. It is vital for living organisms. These processes either increase or decrease the blood glucose concentration but they work together in order to maintain an optimal level.
Glucose is derived from carbohydrates taken in the diet. Carbohydrate is digested to the simple sugars: glucose, fructose and galactose. These sugars are absorbed in the intestine and transported to the liver via the portal vein. Thereafter the liver converts fructose and galactose into glucose. Rising levels of glucose in the blood stimulate the release of insulin from the b cells of the islets of Langerhans in the pancreas.
Insulin is the only hormone that reduces blood glucose levels, and it does this by activating the glucose transport mechanisms and glucose-utilizing metabolic pathways in different tissues of the body. Thus insulin down-regulates glucose forming pathways.
Insulin stimulates:-
uptake of glucose by muscle and adipose tissue
glycolysis
glycogenesis (formation of glycogen from free glucose)
protein synthesis
Insulin inhibits:-
gluconeogenesis (formation of glucose from aminoacids, fatty acids etc.)
lipolysis (breakdown of fatty acids)
proteolysis (breakdown of proteins)
ketogenesis (formation of ketone bodies)
BRAIN
- Very active respiratory metabolism: ~20% of the body’s total oxygen consumption and ~60% of our daily intake of glucose, used primarily to maintain the Na+ / K+ gradient across neuronal membranes.
- The brain cannot store glycogen.
- It also cannot use fatty acids as fuels, since albumin can’t cross the blood brain barrier.
- It can switch to ketone bodies when necessary to minimize protein degradation.
SKELETAL MUSCLES.
- Can use fatty acids, glucose, and ketone bodies as fuel. It has large glycogen stores but uses them solely for itself, never exports (it lacks glucose 6 phosphatase unlike the liver).
- Muscle can be divided into resting, moderately active and active. Resting muscle mostly uses fatty acids as fuel. Moderately active muscle uses glucose from glycogen as well as fatty acids. Active muscle runs glycolysis at a rate exceeding the rate of the CAC, resulting in lactate buildup. Lactate is later converted back to glucose in the Cori Cycle.
- The muscle produces a lot of alanine through transamination of pyruvate. See alanine-glucose cycle.
- During active exercise, pre-existing ATP stores are gone within 5 seconds. Then phosphocreatine kicks in and can last 10-15 seconds. Then anaerobic metabolism kicks in for 45-80 seconds, converting pyruvate to lactate. However this drops the pH in the muscle and is therefore unsustainable – why you can’t sprint for a long time. After ~80 seconds, you go to aerobic exercise, in which you’re generating ATP using the ETC, which is slow but efficient.
- Almost exclusively aerobic, with a high density of mitochondria. Fatty acids are the primary fuel source but they can also use glucose, ketone bodies and lactate.
- Carbohydrates : The liver acts as a blood glucose buffer, takes up and releases glucose into the blood via GLUT2. G6P in the liver has three fates: glycogen production, glycolysis or the pentose phosphate pathway. The liver creates glucose from glycogen breakdown and gluconeogenesis.
- Lipids : When fuel supplies are ample, the liver synthesizes fatty acids. It releases fatty acids it has synthesized or that have been liberated from adipose tissue as VLDLs. During starvation, it converts fatty acids to ketone bodies.
- Amino acids : The liver absorbs most of the dietary amino acids. It can either synthesize proteins or catabolize amino acids depending on metabolic needs. It runs the urea cycle when needed to remove nitrogen.
- When fuel is ample, stores fatty acids as TAGs from VLDL and chlyomicrons.
- When demand for fuel increases, hormone-sensitive lipases (activated by glucagon and epinephrine) mobilize fatty acids.
- Is also an important endocrine organ.
METABOLIC CONTROL BY HORMONE
The metabolic pathways are complex and interdependent. With the changing environments the reactions of metabolism must be finely regulated to maintain a constant set of conditions within cells, a condition called homeostasis. Control of metabolic pathways also allows organisms to respond to signals and interact actively with their environments.
Control of metabolism
Coarse Control: control of the amount of an enzyme. This is a slow process as it involves protein
synthesis.
Fine Control: control of the activity of the enzyme. This is a fast process as it involves changing
the activity of enzyme already available in the cells.
For example :, the metabolic control of carbohydrate.
Glucose homeostasis is a complicated interaction of metabolic pathways. It is vital for living organisms. These processes either increase or decrease the blood glucose concentration but they work together in order to maintain an optimal level.
Glucose is derived from carbohydrates taken in the diet. Carbohydrate is digested to the simple sugars: glucose, fructose and galactose. These sugars are absorbed in the intestine and transported to the liver via the portal vein. Thereafter the liver converts fructose and galactose into glucose. Rising levels of glucose in the blood stimulate the release of insulin from the b cells of the islets of Langerhans in the pancreas.
Insulin is the only hormone that reduces blood glucose levels, and it does this by activating the glucose transport mechanisms and glucose-utilizing metabolic pathways in different tissues of the body. Thus insulin down-regulates glucose forming pathways.
Insulin stimulates:-
uptake of glucose by muscle and adipose tissue
glycolysis
glycogenesis (formation of glycogen from free glucose)
protein synthesis
Insulin inhibits:-
gluconeogenesis (formation of glucose from aminoacids, fatty acids etc.)
lipolysis (breakdown of fatty acids)
proteolysis (breakdown of proteins)
ketogenesis (formation of ketone bodies)
METABOLIC RESPONSES TO STRESS CONDITION.
The metabolic response to stress is part of the adaptive response to survive critical illness. Several mechanisms are well preserved during evolution, including the stimulation of the
sympathetic nervous system, the release of pituitary hormones, a peripheral resistance to the effects of these and other anabolic factors, triggered to increase the provision of energy substrates to the vital tissues.
The pathways of energy production are altered and alternative substrates are used as a result of the loss of control of energy substrate utilization by their availability. The clinical consequences of the metabolic response to stress include sequential changes in energy expenditure, stress hyperglycaemia, changes in body composition, and psychological and behavioural problems.
The loss of muscle proteins and function is a major long-term consequence of stress metabolism. Specific therapeutic interventions, including hormone supplementation, enhanced protein intake, and early mobilization, are investigated. This review aims to summarize the pathophysiological mechanisms, the clinical consequences, and therapeutic implications of the metabolic response to stress.
BIOCHEMICAL FACTORS IN OBESITY.
Obesity is one of the major risk factors of type 2 diabetes and is associated with many metabolic dearangements that impair insulin sensitivity. These abnormalities include excess lipolysis causing increased concentrations of non-esterified fatty acids and triglycerides in blood and skeletal muscle. Muscle glucose uptake is suppressed.
Figure below gives the possible biochemical mechanisms involved in the evolution of insulin resistance and insulin-resistant states including diabetes.
Figure below gives the possible biochemical mechanisms involved in the evolution of insulin resistance and insulin-resistant states including diabetes.
Obesity is reported to affect insulin action by changing the secretion of adipocytokines, specifically of leptin and adiponectin and leads to proinflammatory conditions. Features of insulin resistance, including hypertriglyceridaemia and increased abdominal or visceral fat, are seen even in non-obese populations.
Insulin resistance is associated with visceral and subcutaneous fat content . Glucose disposal rate and plasma adiponectin concentration are inversely related to fat-cell size. Fatty acid influx to the liver is an important pathogenetic factor for fatty liver and is also a determinant of excess triglyceride-rich lipoproteins. Dyslipidaemia in type 2 diabetes is more severe in the presence of fatty liver.
Insulin resistance is associated with visceral and subcutaneous fat content . Glucose disposal rate and plasma adiponectin concentration are inversely related to fat-cell size. Fatty acid influx to the liver is an important pathogenetic factor for fatty liver and is also a determinant of excess triglyceride-rich lipoproteins. Dyslipidaemia in type 2 diabetes is more severe in the presence of fatty liver.